Patients grow tired of the multiple eye injections required to treat wet macular degeneration. These injections can be anxiety-provoking, uncomfortable, and as frequent as every month. Fortunately, help is on the way. A new FDA-approved treatment, Susvimo, has arrived that is indicated for twice-per-year dosing. In addition to this innovative product, a new injectable medication, Vabysmo, has gained FDA approval for dosing up to 16 weeks. Lastly, there are other medications being assessed in clinical trials that may soon provide longer-lasting dosing than what is currently available.
It Is Worth Saying, Eye Injections Are a Miracle
Eye injections for age related macular degeneration have been around since the early 2000’s. Originally, anti-vascular endothelial growth factor (anti-VEGF) medication was designed to inhibit the growth of cancer, but it was quickly proven to stop and reverse vision loss from wet macular degeneration. These miracle drugs have provided the greatest prevention of blindness since the advent of cataract surgery. Patients who would have been blind twenty years ago have maintained reading and driving vision thanks to these wonderful medications.
The Problem of Frequent Eye Injections
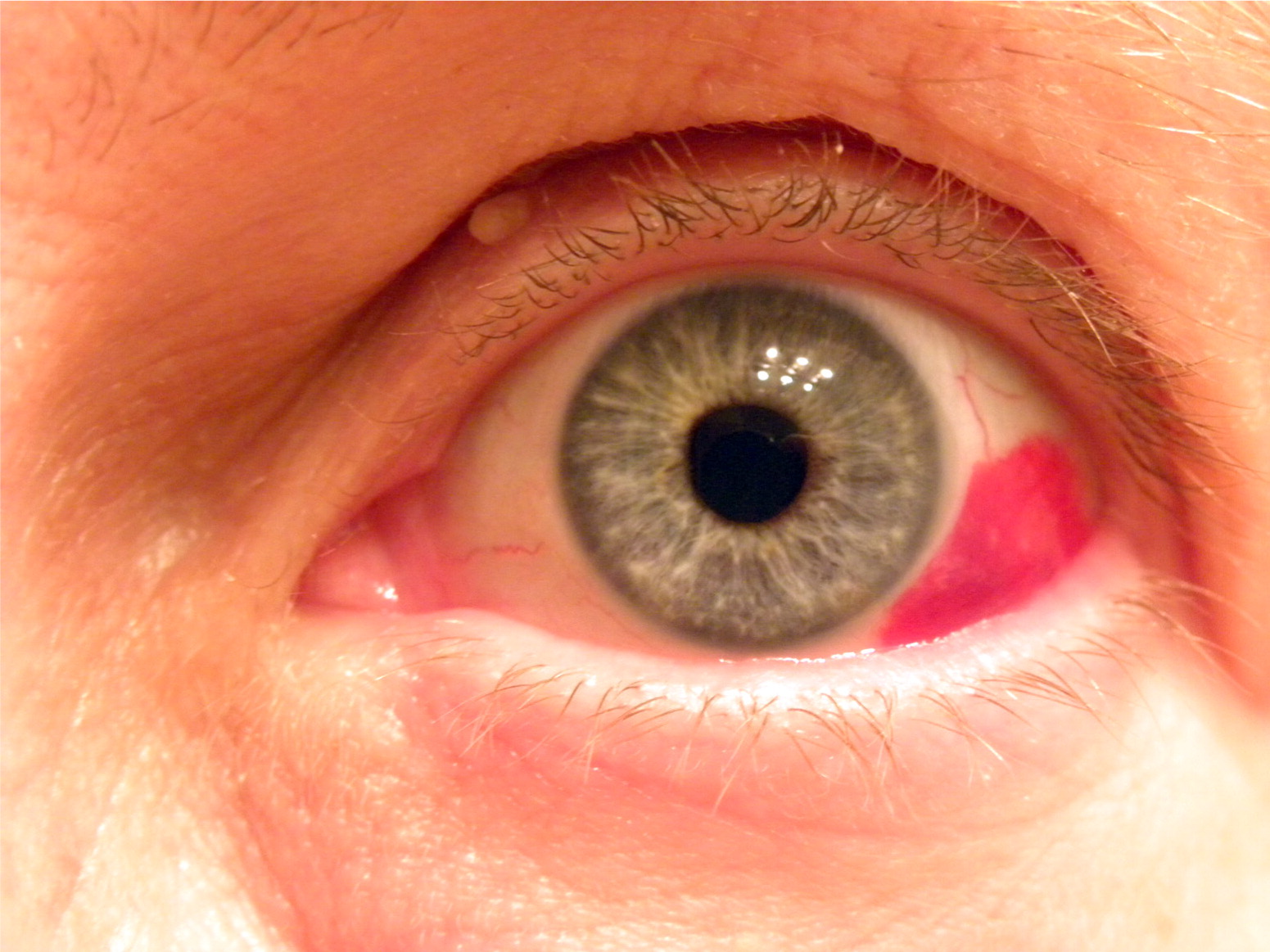
Many patients with wet macular degeneration are required to get injections of medication into their eye every 4-6 weeks. These frequent visits can be stressful on the schedule and the psyche of macular degeneration patients. A typical injection visit lasts 45-minutes to an hour and sometimes involves dilation, retinal imaging, and multiple-drop preparation in advance of the eye injection. The injection itself can sometimes be uncomfortable or cause bleeding on the surface of the eye. Afterwards, some patients feel scratchy or irritated for the rest of the day. The process gets repeated every visit indefinitely, with some patients receiving greater than fifty injections in their lifetime. Oftentimes, life events prevent patients from coming to their appointments on schedule, which results in loss of vision over time.
Injection Frequency Has Been Extending
Ever since Lucentis was approved for monthly dosing in 2006, physicians have been looking for ways to perform injections less-frequently while maintaining visual acuity gains. Initially, an as-needed dosing regimen was attempted, where patients only received medication for recurrences of macular leakage.
Unfortunately, this approach was found to cause loss of vision over time compared to monthly dosing. A more conservative paradigm was developed for decreasing injection frequency, called ‘Treat and Extend.’ In this method, injection visits are slowly spread out until leakage occurs, at which point the injection interval is maintained at the maximum-extended interval. This has been shown to achieve similar vision gains to monthly treatments, but with fewer injections.
Another way that injection frequency has been extended is by the development of more powerful anti-VEGF medication. Eylea was approved for 8-week dosing in 2011, and then for 12-week dosing in 2018. Another medication, Bevou, was approved for 12-week dosing in 2019, but fell out of favor due to a higher rate of ocular inflammation.
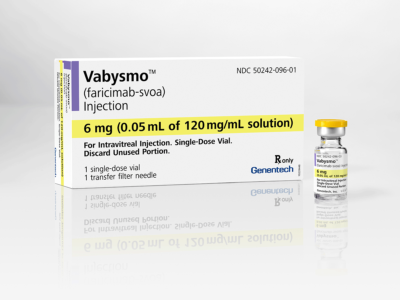 A new injectable medication, Vabysmo, has recently gained FDA approval for up to 16-week dosing. This novel treatment blocks VEGF as well as a second chemical signal for new blood vessel growth called ANG-2. Vabysmo will be available this year to patients with both wet macular degeneration and diabetic retinopathy.
A new injectable medication, Vabysmo, has recently gained FDA approval for up to 16-week dosing. This novel treatment blocks VEGF as well as a second chemical signal for new blood vessel growth called ANG-2. Vabysmo will be available this year to patients with both wet macular degeneration and diabetic retinopathy.
Other attempts at extending injection intervals have been unsuccessful, include retinal laser, focal radiation, eye drops, and oral medication.
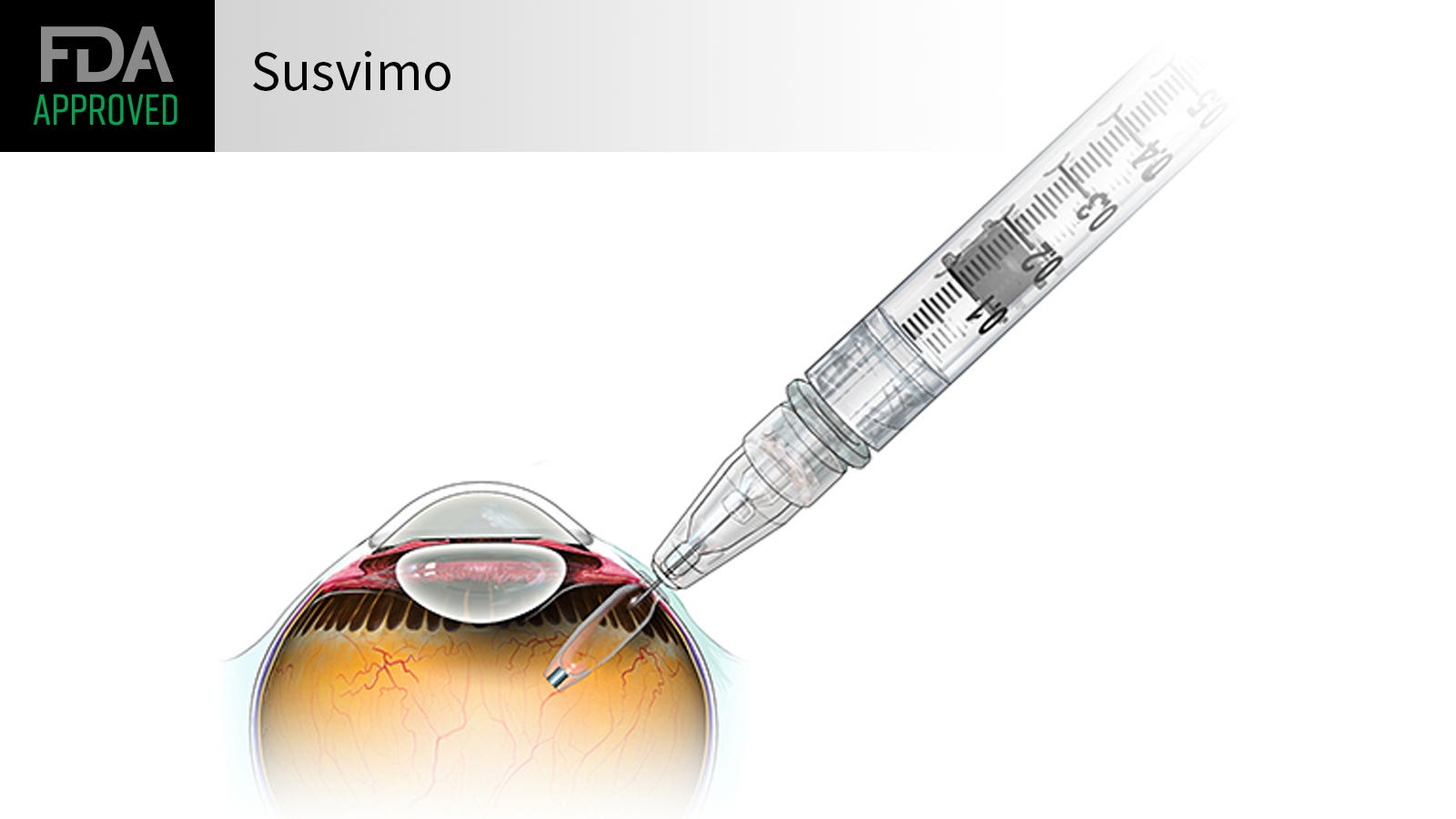
Susvimo: FDA Approved Eye Injections that last up to 6 months
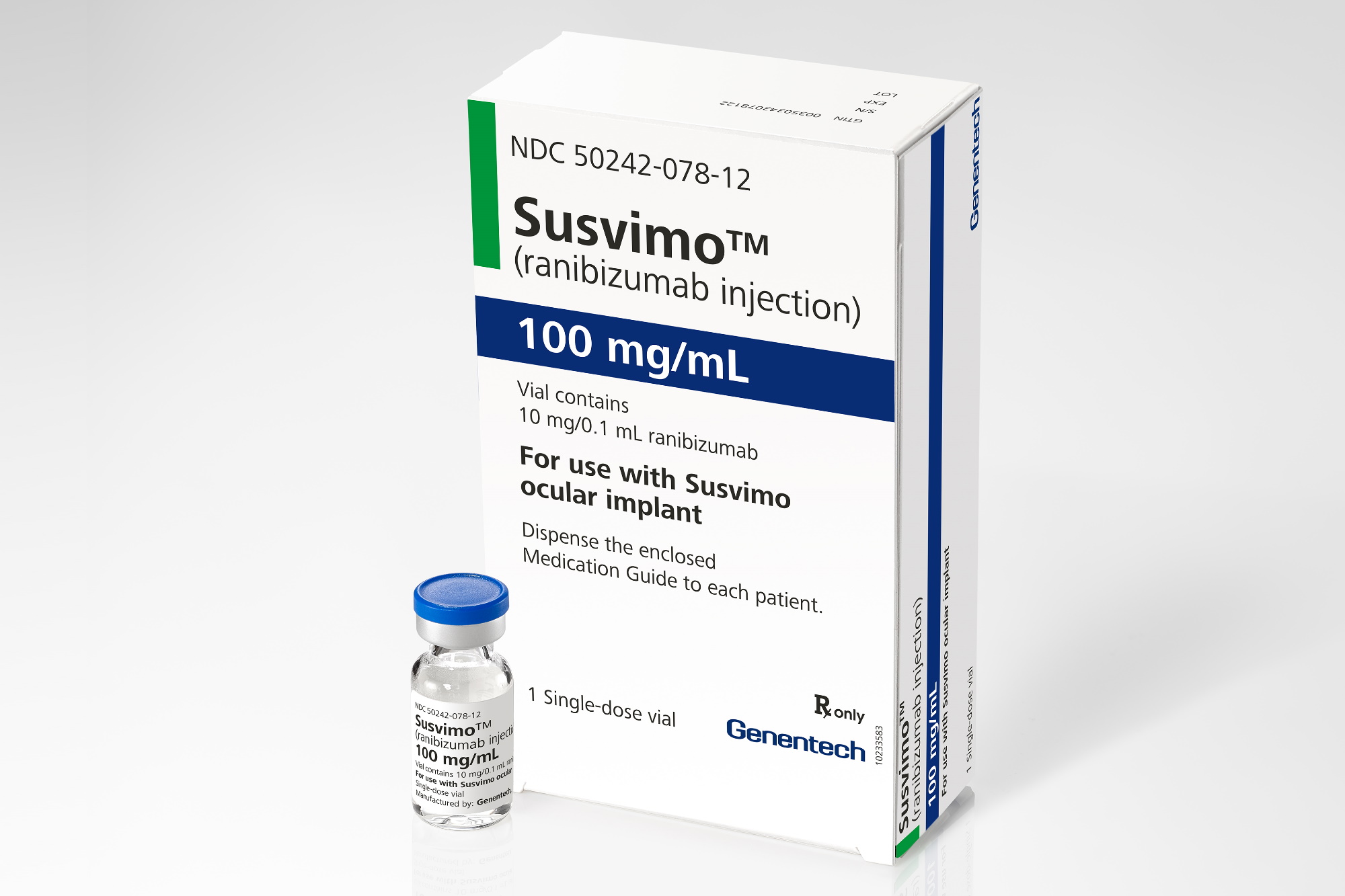
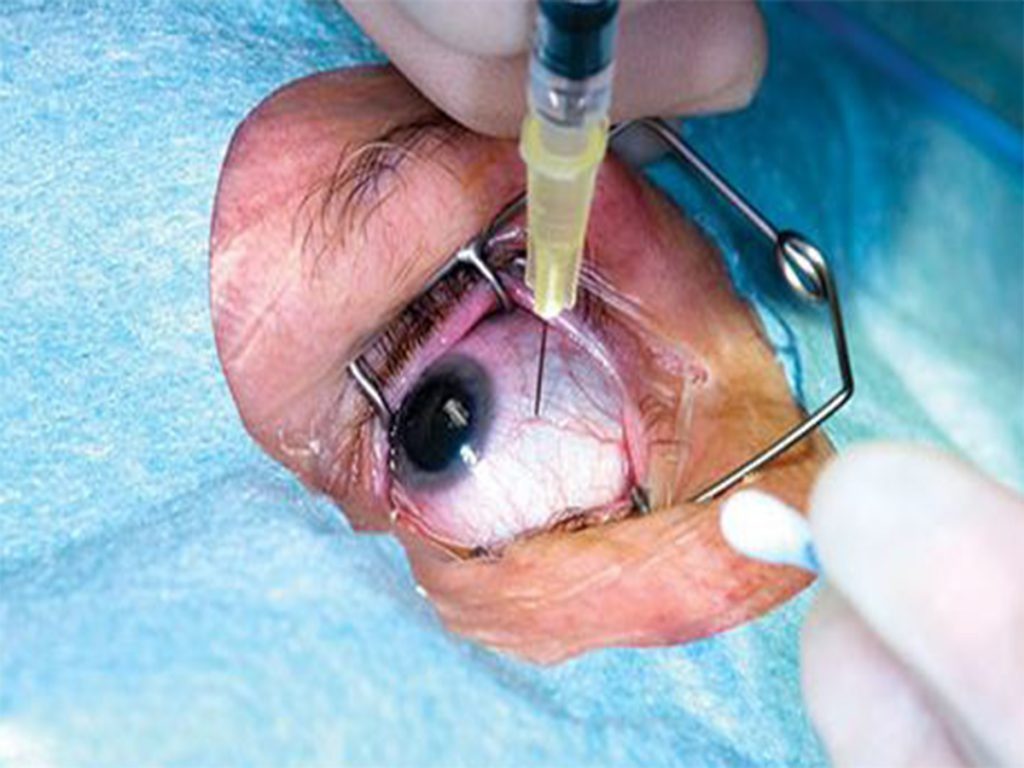
Doctors and scientists have been searching for additional ways to extend eye injection intervals. One method that just received FDA-clearance is Susvimo, which is dosed with an injection in the clinic every 6 months. Susvimo is an intraocular implant that is placed within the eye through an incision and covered with the thin membrane on the surface of the eye called the conjunctiva. It is pre-loaded with medication and delivers a steady-state of drug for at least 6 months, with less than 2% of patients requiring another injection prior to that time. At the end of 6 months, the implant is refilled with an injection in the clinic.
The main risk of Susvimo is post-operative infection, which occurred in 2% of patients during the clinical trial. Infection in the eye is a serious threat to vision, and careful coverage of the implant with the conjunctiva is necessary to reduce the risk of this complication.
Who is Susvimo for?
The best candidates for Susvimo implantation are those who are currently receiving eye injections every 4-6 weeks. The risk/benefit profile of this procedure favors those with better vision in their non-operative eye. Patients who only get injections in one eye are also favored given that they will only need one implant procedure to see the benefits of less-frequent eye injection appointments. Those who have difficulty making frequent appointments due to their schedule, disability, or lack of transportation are obvious winners with Susvimo.
I am very impressed with the friendliness of the office staff and how well the office is run. There was not a long wait in between room visits. I am also impressed with the modern equipment they use and especially impressed with the competence of Dr. Shane and how well he explained everything.
Other Opportunities to Extend Eye Injection Intervals
Susvimo is not the last innovation that will allow retina specialists to extend eye injection intervals. Several drugs are in clinical trials that are more potent VEGF inhibitors than the current FDA-approved medications. Other investigational drugs target molecules along the wet macular degeneration pathway other than VEGF.
Most exciting in the research of wet macular degeneration treatment is gene therapy. While gene therapy trials are in their preliminary stages, this class of medication has the potential to prevent wet macular degeneration with one injection. These drugs accomplish long-lasting dosing by infecting retina cells with the gene for producing aflibercept (Eylea). Once infected, a retinal cell has the ability to produce a steady-state of anti-VEGF medication indefinitely. Early trials have indicated that 60% of patients injected with gene therapy were able to go at least one year without experiencing a recurrence of macular edema.
Call Us to Explore Options for Less-Frequent Eye Injections
Dr. Shane offers all of the latest FDA-approved options for less-frequent eye injections, including Susvimo. If you have wet macular degeneration and would like to explore how to get fewer injections while maintaining your vision, call or email us for a consultation. We are here to help you!
